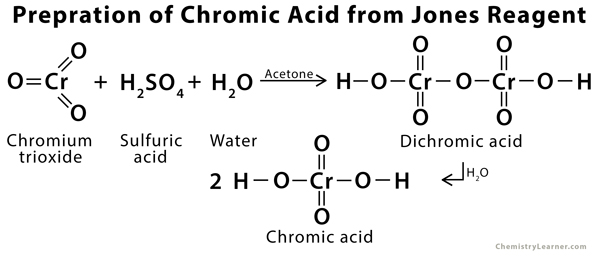
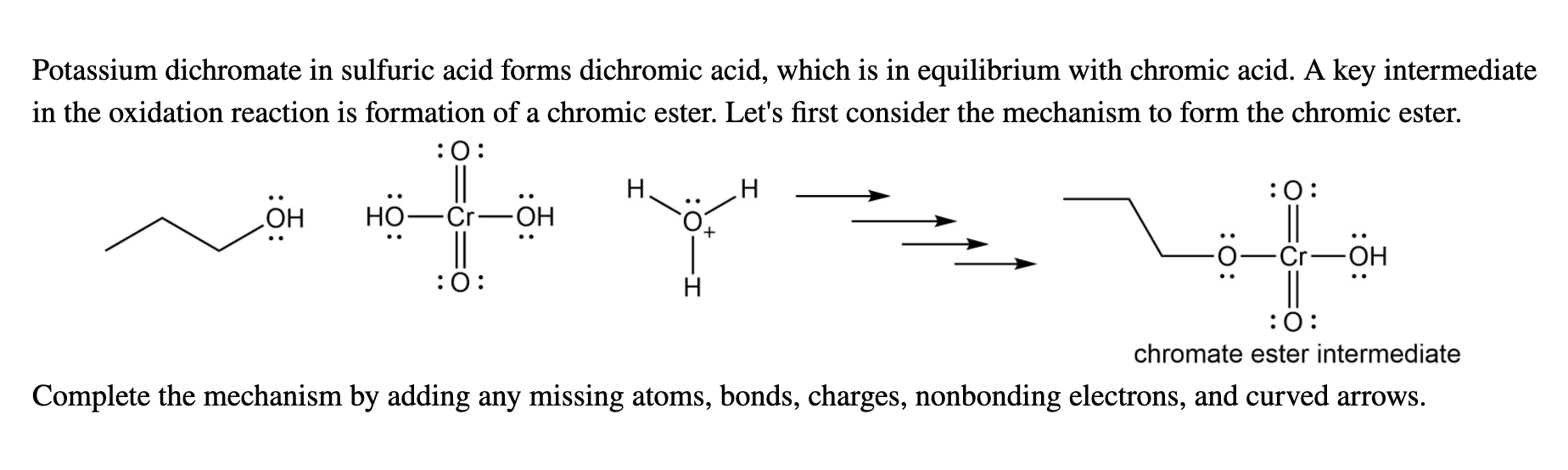
Periodic TABLE Elements - Chromic acid The term chromic acid is usually used for a mixture made by adding concentrated sulfuric acid to a dichromate, which may contain a variety of compounds,
Dichromic acid, (H2Cr2O7), disodium salt, dihydrate - Chemical Details | Chemical Search | Pesticides | US EPA

SOLVED: What is the proper name for H2CrO4 (aq)? hydrogen chromate chromium acid chromic acid hydrochromic acid dichromic acid
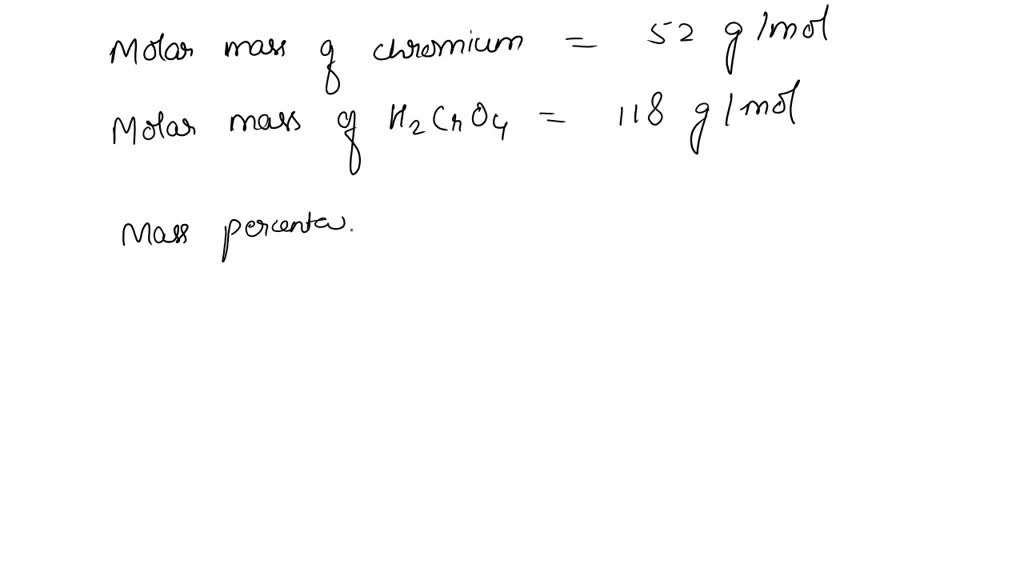
SOLVED: Which substance has the greater mass percent of chromium: chromic acid, H2CrO4(aq), or dichromic acid, H2Cr2O7(aq)?
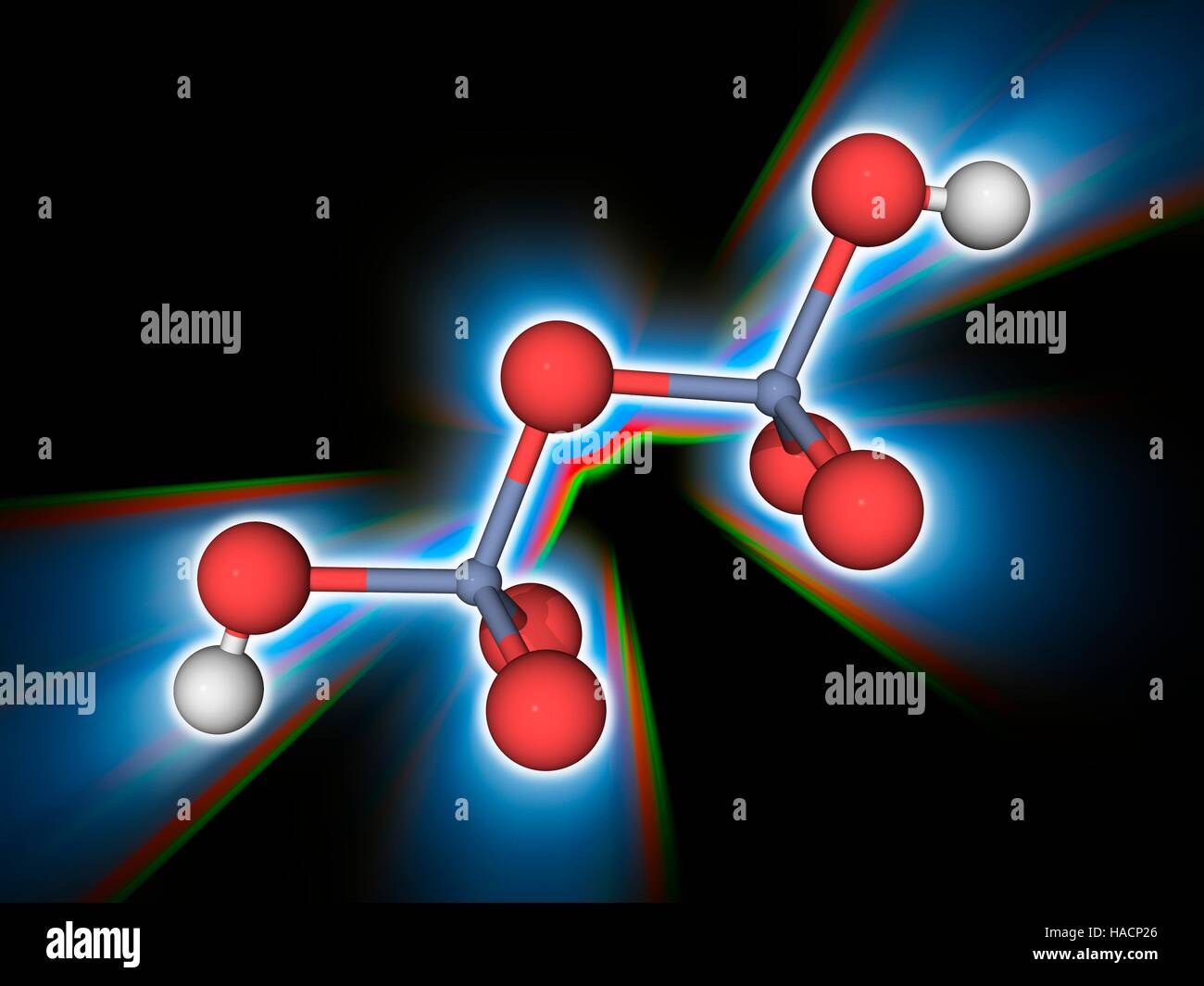
Dichromic acid. Molecular model of the strong acid and corrosive oxidizing agent dichromic acid (H2.Cr2.O7). Formed in mixtures of chromic acid, this is the fully protonated form of the dichromate ion. Uses